Exploring the therapeutic potential of honeybee venom in mitigating diabetic reproductive complications: insights from in vitro and in vivo studies in rats
DOI:
https://doi.org/10.5380/avs.v31i1.101661Palavras-chave:
Honeybee venom, diabetes, oxidative stress, sperm motility, antioxidant activity.Resumo
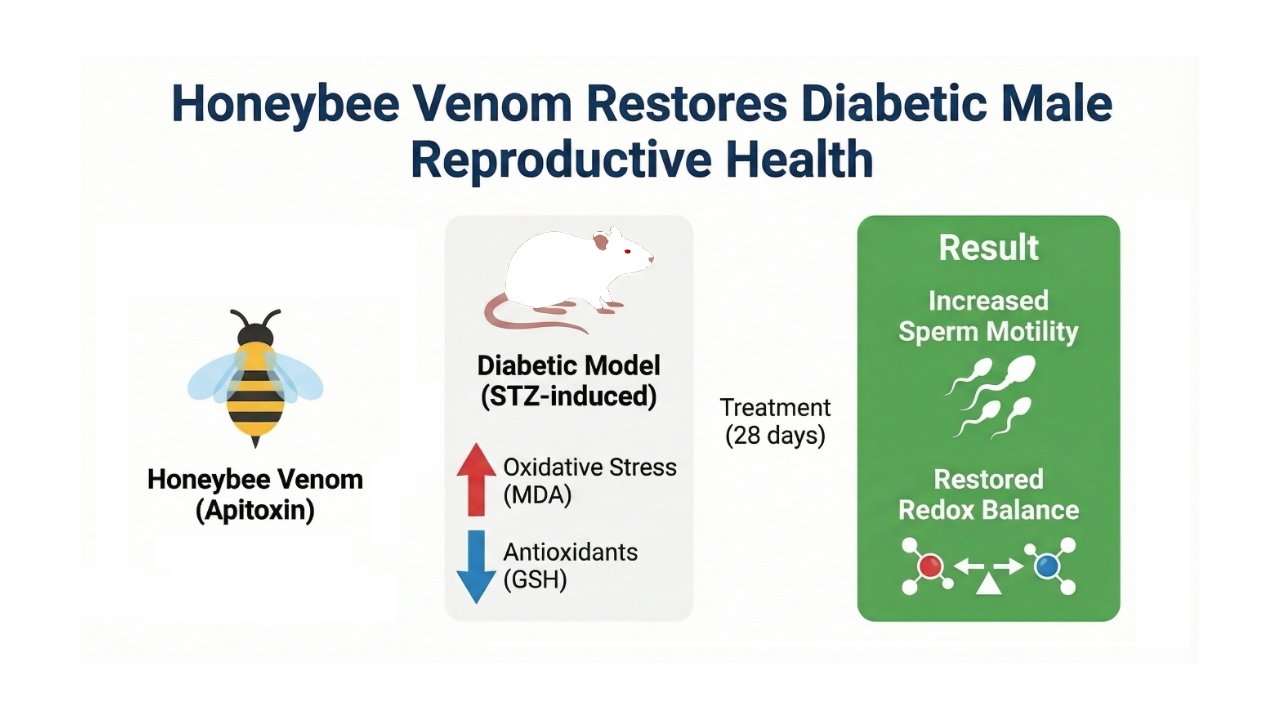
Diabetes mellitus impairs male reproductive health through oxidative stress–mediated pathways. This study investigated the antioxidant capacity and reproductive effects of honeybee venom (apitoxin) in diabetic male rats. The in vitro antioxidant activity of bee venom showed a DPPH radical scavenging rate of 76.85 ± 1.55%, close to that of ascorbic acid (79.29 ± 1.82%, P < 0.001), and a metal chelating activity of 91.69 ± 2.55% (P = 0.227). For in vivo evaluation, streptozotocin-induced diabetic Wistar rats (n = 6/group) were assigned to control (C), apitoxin-treated (A), diabetic (D), and diabetic + apitoxin (DA) groups for 28 days. Diabetic rats exhibited markedly decreased sperm motility (61.4 ± 6.9%) compared to C (78.6 ± 3.8%) and A (82.9 ± 4.9%) (P = 0.007), whereas apitoxin treatment in DA rats improved motility to 77.1 ± 7.6%. Epididymal, vesicular, and prostatic weights were reduced in diabetic groups (P < 0.01). Oxidative stress was evident with elevated MDA in D (49.3 ± 31.1 nmol/mg protein) versus DA (27.7 ± 4.4) and C (29.0 ± 3.0) (P = 0.023), while GSH was significantly higher in DA (54.4 ± 18.4 μmol/mg protein) compared to D (35.2 ± 4.2) (P = 0.006). Overall, honeybee venom ameliorated diabetes-induced oxidative damage and improved sperm quality through redox modulation. These findings highlight its potential as a natural therapeutic agent against diabetic reproductive complications.
Referências
Abdelhamid MS, El Bohi KM, Sherif MH, et al. Bee venom ameliorates methyl mercury-induced reproductive impairment in male Sprague Dawley rats. Biochemistry Lett, 19:(1);138–150, 2023. (DOI: https://doi.org/10.21608/blj.2025.231715.1045)
Al-Hatamleh MA, Boer JC, Wilson KL, et al. Antioxidant-based medicinal properties of stingless bee products: Recent progress and future directions. Biomolecules, 10:(6);923, 2020. (DOI: https://doi.org/10.3390/biom10060923)
Atta MS, Almadaly EA, El-Far AH, et al. Thymoquinone defeats diabetes-induced testicular damage in rats targeting antioxidant, inflammatory and aromatase expression. Int J Mol Sci, 18:(5);919, 2017. (DOI: https://doi.org/10.3390/ijms18050919)
Aufschnaiter A, Kohler V, Khalifa S, et al. Apitoxin and its components against cancer, neurodegeneration and rheumatoid arthritis: Limitations and possibilities. Toxins, 12:(2);66, 2020. (DOI: https://doi.org/10.3390/toxins12020066)
Badejogbin OC, Chijioke-Agu OE, Olubiyi MV, et al. Pathogenesis of testicular dysfunction in diabetes: exploring the mechanism and therapeutic interventions. J Assist Reprod Genet, 42:(2);367–379, 2025. (DOI: https://doi.org/10.1007/s10815-024-03314-3)
Cortas NK, Wakid NW. Determination of inorganic nitrate in serum and urine by a kinetic cadmium-reduction method. Clin Chem, 36:(8);1440–1443, 1990. (DOI: https://doi.org/10.1093/clinchem/36.8.1440)
Denk B, Fidan AF, et al. Effects of honeybee (Apis mellifera) venom on redox balance, biochemical and hematological profile in diabetic rats: A preliminary study. Türk J Vet Anim Sci, 45:(2);257–265, 2021. (DOI: https://doi.org/10.3906/vet-2006-139)
Denk B. Exploring Apis mellifera L. Venom’s Antioxidant Power in Various Solvents: Unveiling its In vitro Potential. Kocatepe Vet J, 16:(3);420–431, 2023. (DOI: https://doi.org/10.30607/kvj.1343130)
Dontha S. A review on antioxidant methods. Asian J Pharm Clin Res, 9:(2);14–32, 2016. (DOI: http://dx.doi.org/10.22159/ajpcr.2016.v9s2.13092)
Du Sert NP, Ahluwalia A, Alam S, et al. Reporting animal research: Explanation and elaboration for the ARRIVE guidelines 2.0. PLoS biology, 18:(7);e3000411, 2020. (DOI: https://doi.org/10.1371/journal.pbio.3000411)
El-Hanoun A, El-Komy A, El-Sabrout K, et al. Effect of bee venom on reproductive performance and immune response of male rabbits. Physiol Behav, 223:(1);112987, 2020. (DOI: https://doi.org/10.1016/j.physbeh.2020.112987)
Elkomy A, El-Hanoun A, Abdella M, et al. Improving the reproductive, immunity and health status of rabbit does using honey bee venom. J Anim Physiol Anim Nutr, 105:(5);975–983, 2021. (DOI: https://doi.org/10.1111/jpn.13552)
Frangieh J, Salma Y, Haddad K, et al. First characterization of the venom from Apis mellifera syriaca, a honeybee from the Middle East region. Toxins, 11:(4);191, 2019. (DOI: https://doi.org/10.3390/toxins11040191)
Furman BL. Streptozotocin‐induced diabetic models in mice and rats. Curr Protoc Pharmacol, 70:(1);5–47, 2015. (DOI: https://doi.org/10.1002/0471141755.ph0547s70)
Gandhi J, Dagur G, Warren K, et al. The role of diabetes mellitus in sexual and reproductive health: an overview of pathogenesis, evaluation, and management. Curr Diabetes Rev, 13:(6);573–581, 2017. (DOI: https://doi.org/10.2174/1573399813666161122124017)
Gulcin İ, Alwasel SH. Metal ions, metal chelators and metal chelating assay as antioxidant method. Processes, 10:(1);132, 2022. (DOI: https://doi.org/10.3390/pr10010132)
Güngör Ş, Yeni D, İnanç M, et al. Evaluation of the in vitro cryopreservative performance of Juniper berry oil (Juniperus communis) on frozen-thawed bull semen. Acta Vet Brno, 92:(4);335-342, 2023. (DOI: http://doi.org/10.2754/avb202392040335)
Hwang YY, Tsen QT, Felim J, et al. Probiotics as a therapeutic approach to alleviate reproductive harm from polystyrene microplastics in male rats. Sci Rep, 15:(1);34783, 2025. (DOI: https://doi.org/10.1038/s41598-025-18550-5)
Kasozi KI, Niedbała G, Alqarni M, et al. Bee venom—a potential complementary medicine candidate for SARS-CoV-2 infections. Front Public Health, 8:(1);594458, 2020. (DOI: https://doi.org/10.3389/fpubh.2020.594458)
Keshta AT, Fathallah AM, Attia YA, et al. Ameliorative effect of selenium nanoparticles on testicular toxicity induced by cisplatin in adult male rats. Food Chem Toxicol, 179:(1);113979, 2023. (DOI: https://doi.org/10.1016/j.fct.2023.113979)
Khoei HH, Fakhri S, Parvardeh S, et al. Testicular toxicity and reproductive performance of streptozotocin-induced diabetic male rats: the ameliorating role of silymarin as an antioxidant. Tox Rev, 38:(3);223-233, 2019. (DOI: https://doi.org/10.1080/15569543.2018.1444641)
Korac B, Kalezic A, Pekovic-Vaughan V, et al. Redox changes in obesity, metabolic syndrome, and diabetes. Redox Biol. 42; 101887, 2021. (DOI: https://doi.org/10.1016/j.redox.2021.101887)
Leisegang K, et al. Oxidative stress in men with obesity, metabolic syndrome and type 2 diabetes mellitus: Mechanisms and management of reproductive dysfunction. In: Oxidative Stress and Toxicity in Reproductive Biology and Medicine: A Comprehensive Update on Male Infertility-Volume One. Cham: Springer International Publishing, 237–256, 2022. (DOI: https://doi.org/10.1007/978-3-030-89340-8_11)
Li H, Huo Y, He X, et al. A male germ-cell-specific ribosome controls male fertility. Nature, 612:(7941);725–731, 2022. (DOI: https://doi.org/10.1038/s41586-022-05508-0)
Minas A, Camargo M, Alves MG, et al. Effects of diabetes-induced hyperglycemia on epigenetic modifications and DNA packaging and methylation during spermatogenesis; A narrative review. Iran J Basic Med Sci, 27:(1);3, 2024. (DOI: https://doi.org/10.22038/IJBMS.2023.69604.15173)
Moradi M, Jahromi MG, Mirzaei S, et al. Protective Effects of Scrophularia striata Extract on Testicular Function and Spermatogenesis in Cadmium‐Exposed Mice: Role of Nitro‐Oxidative Stress Modulation. Andrologia, 2024:(1);5565361, 2024. (DOI: https://doi.org/10.1155/and/5565361)
Mousavi SM, Imani S, Haghighi S, et al. Effect of Iranian honey bee (Apis mellifera) venom on blood glucose and insulin in diabetic rats. J Arthropod-Borne Dis, 6:(2);136, 2012.
Munteanu IG, Apetrei C. Analytical methods used in determining antioxidant activity: A review. Int J Mol Sci, 22:(7);3380, 2021. (DOI: https://doi.org/10.3390/ijms22073380)
Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem, 95:(2);351–358, 1979. (DOI: https://doi.org/10.1016/0003-2697(79)90738-3)
Pinto D, Vieira EF, Peixoto AF, et al. Optimizing the extraction of phenolic antioxidants from chestnut shells by subcritical water extraction using response surface methodology. Food Chem. 334; 127521, 2021. (DOI: https://doi.org/10.1016/j.foodchem.2020.127521)
Rosenfeld RM, Grega ML, Karlsen MC, et al. Lifestyle interventions for treatment and remission of type 2 diabetes and prediabetes in adults: a clinical practice guideline from the American College of Lifestyle Medicine. Am J Lifestyle Med, 19:(2_suppl);10S–131S, 2025. (DOI: https://doi.org/10.1177/15598276251325488)
Sadeghi R, Fang F, Shao Y, et al. Eliminating protein interference when quantifying potato reducing sugars with the miniaturized Somogyi-Nelson assay. Food Chem, 373:(1);131473, 2022. (DOI: https://doi.org/10.1016/j.foodchem.2021.131473)
Saka WA, Adeogun AE, Adisa VI, et al. L-arginine attenuates dichlorvos-induced testicular toxicity in male Wistar rats by suppressing oxidative stress-dependent activation of caspase 3-mediated apoptosis. Biomed Pharmacother, 178:(1);117136, 2024. (DOI: https://doi.org/10.1016/j.biopha.2024.117136)
Sharma S, Mathew AB, Chugh J. miRNAs: Nanomachines that micromanage the pathophysiology of diabetes mellitus. Adv Clin Chem, 82;199–264, 2017. (DOI: https://doi.org/10.1016/bs.acc.2017.06.003)
Sivri D, Akdevelioğlu Y. Effect of fatty acids on glucose metabolism and type 2 diabetes. Nutr Rev, 83:(5);897–907, 2025. (DOI: https://doi.org/10.1093/nutrit/nuae165)
Soudamani S, Yuvaraj S, Malini T, et al. Experimental diabetes has adverse effects on the differentiation of ventral prostate during sexual maturation of rats. Anat Rec A, 287:(2);1281–1289, 2005. (DOI: https://doi.org/10.1002/ar.a.20250)
Suleiman JB, Bakar ABA, Mohamed M. Review on bee products as potential protective and therapeutic agents in male reproductive impairment. Molecules, 26:(11);3421, 2021. (DOI: https://doi.org/10.3390/molecules26113421)
Szabo C. Role of nitrosative stress in the pathogenesis of diabetic vascular dysfunction. Br J Pharmacol, 156:(5);713–727, 2009. (DOI: https://doi.org/10.1111/j.1476-5381.2008.00086.x)
Tietze F. Enzymic method for quantitative determination of nanogram amounts of total and oxidized glutathione: applications to mammalian blood and other tissues. Anal Biochem, 27:(3);502–522, 1969. (DOI: https://doi.org/10.1016/0003-2697(69)90064-5)
Tousoulis D, Kampoli A-M, Tentolouris N, et al. The role of nitric oxide on endothelial function. Curr Vasc Pharmacol, 10:(1);4–18, 2012. (DOI: https://doi.org/10.2174/157016112798829760)
Yeni D, Güngör Ş, Avdatek F, et al. Investigation of changes in spermatozoon characteristics, chromatin structure, and antioxidant/oxidant parameters after freeze-thawing of hesperidin (vitamin P) doses added to ram semen. Life, 12:(11);1780, 2022. (DOI: https://doi.org/10.3390/life12111780)
Yoon S-Y, Kwon Y-B, Kim H-W, et al. Bee venom injection produces a peripheral anti-inflammatory effect by activation of a nitric oxide-dependent spinocoeruleus pathway. Neurosci Lett, 430:(2);163–168, 2008. (DOI: https://doi.org/10.1016/j.neulet.2007.10.035)
Zhang A, Zhichen B, Kidoguchi S, et al. An SGLT2 inhibitor, canagliflozin, reduces blood glucose level in the renal capillaries and protects the capillary network in the diabetic rats. Diabetes Obes Metab, Early view;1–9, 2025. (DOI: https://doi.org/10.1111/dom.70118)
Zhao L, Makinde EA, Olatunji OJ, et al. Protective effects of ethyl acetate extract from Shorea roxburghii against diabetes induced testicular damage in rats. Environ Toxicol, 36:(3);374–385, 2021. (DOI: https://doi.org/10.1002/tox.23043)
Zhu Y, Du Q, Jiao N, et al. Catalpol ameliorates diabetes-induced testicular injury and modulates gut microbiota. Life Sci, 267;118881, 2021. (DOI: https://doi.org/10.1016/j.lfs.2020.118881)
Publicado
Como Citar
Edição
Seção
Licença

Este trabalho está licenciado sob uma licença Creative Commons Attribution-ShareAlike 4.0 International License.
Autores que publicam nesta revista concordam com os seguintes termos:
- Autores mantém os direitos autorais e concedem à revista o direito de primeira publicação, com o trabalho simultaneamente licenciado sob a Creative Commons - Atribuição 4.0 Internacional que permite o compartilhamento do trabalho com reconhecimento da autoria e publicação inicial nesta revista.
- Autores têm autorização para assumir contratos adicionais separadamente, para distribuição não-exclusiva da versão do trabalho publicada nesta revista (ex.: publicar em repositório institucional ou como capítulo de livro), com reconhecimento de autoria e publicação inicial nesta revista.
- Autores têm permissão e são estimulados a publicar e distribuir seu trabalho online (ex.: em repositórios institucionais ou na sua página pessoal) a qualquer ponto antes ou durante o processo editorial, já que isso pode gerar alterações produtivas, bem como aumentar o impacto e a citação do trabalho publicado.













