Dietary supplementation of Yucca schidigera and Gleditsia amorphoides modulates gut fermentation metabolites, antioxidant status, and inflammatory markers in adult dogs
DOI:
https://doi.org/10.5380/avs.v31i1.101516Palavras-chave:
Ammonia, Propionate, Saponin extractsResumo
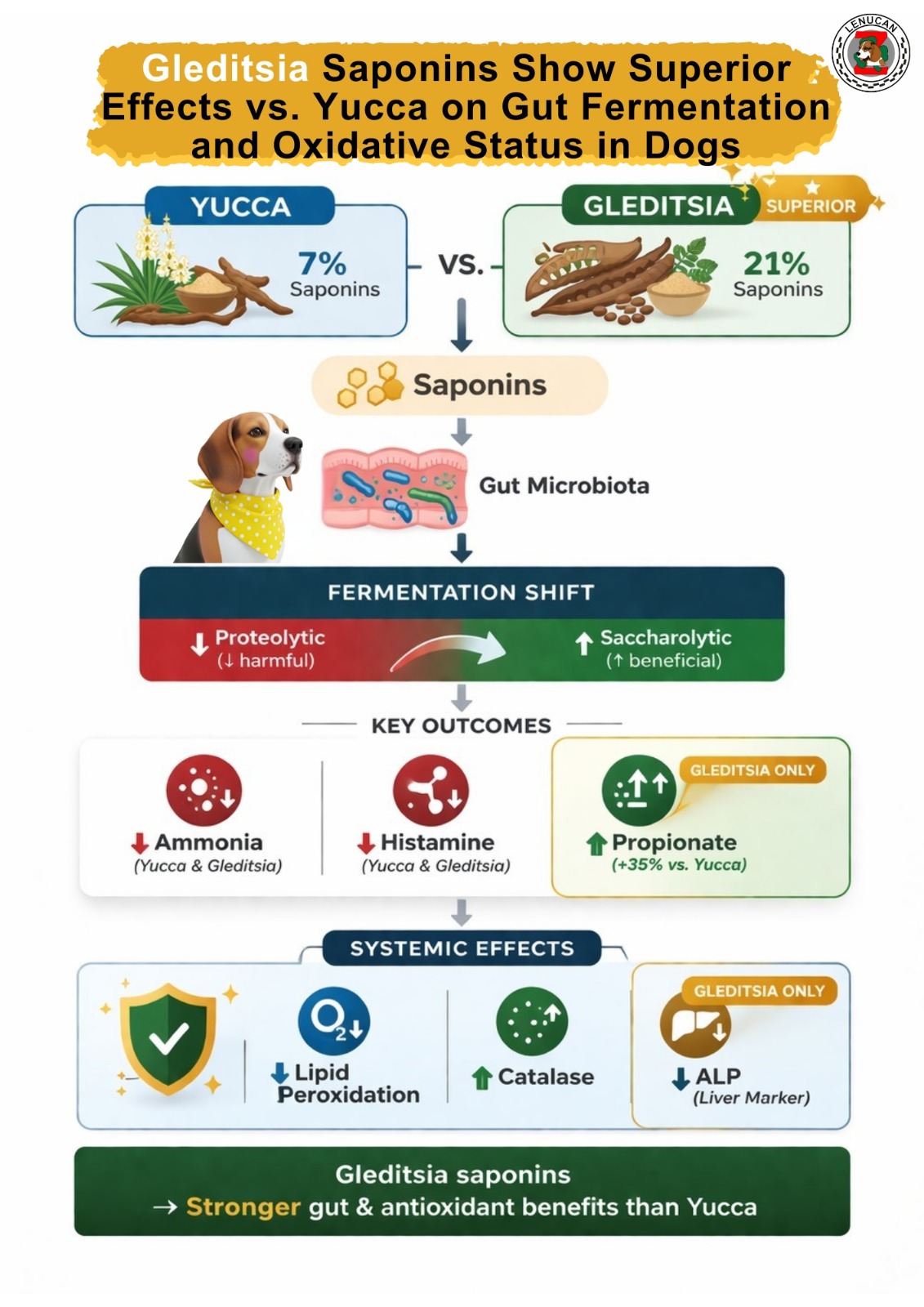
Saponins are bioactive compounds known for modulating potential inflammatory metabolites produced by the gut microbiome, including ammonia. This study evaluated the effects of the saponin sources, Yucca schidigera (7% saponins) and Gleditsia amorphoides (21% saponins), on intestinal functionality, antioxidant, and inflammatory markers in adult dogs. Eighteen Beagle dogs (10 males and eight females), with 12.20 ± 1.33 kg body weight were randomly divided into three groups: Control (no saponins), 200 g Yucca schidigera/ton of diet (Yucca), and 200 g Gleditsia amorphoides/ton of diet (Gleditsia), totaling six replications per treatment. Dogs were fed experimental diets for 20 days. Fresh fecal samples were collected on day 20 to analyze: dry matter, score, pH, ammonia, short-chain fatty acids (SCFA), branched-chain fatty acids (BCFA), and biogenic amines. Blood was also collected on day 20 to analyze inflammatory cytokines, antioxidant enzymes, and liver enzymes. Preliminary results showed that the Gleditsia group had higher fecal propionate concentrations and lower histamine concentrations (P < 0.05). Both the Yucca and Gleditsia groups presented lower fecal concentrations of ammonia and spermine than the Control group (P < 0.05). Additionally, the Yucca and Gleditsia groups showed lower lipid peroxidation and higher catalase activity, whereas only the Gleditsia group showed lower alkaline phosphatase activity than the Control group (P<0.05). In conclusion, Yucca and Gleditsia can modulate fecal fermentative metabolites and improve the antioxidant status of dogs. These findings also showed the safety of using Yucca and Gleditsia during the experimental period.
Key-words: Ammonia, Propionate, Saponin extracts.
Referências
Aebi, H. (1984). Catalase in vitro. Methods in Enzymology. 105, 121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Association of Official Analytical Chemists (AOAC). (1995). Official methods of analysis (16th ed.). AOAC International.
Benzie, I. F. F., Strain, J. J. (1996). The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 239, 70–76. https://doi.org/10.1006/abio.1996.0292.
Bilski, J., Mazur-Bialy, A., Wojcik, D., Zahradnik-Bilska, J., Brzozowski, B., Magierowski, M., Mach, T., Magierowska, K., Brzozowski, T. (2017). The role of intestinal alkaline phosphatase in inflammatory disorders of gastrointestinal tract. Mediators of inflammation. 9074601, 9. https://doi.org/10.1155/2017/9074601
Brito, C. B. M., Félix, A. P., Jesus, R. M., França, M., Oliveira, S. G., Krabbe, E., Maiorka, A. (2010). Digestibility and palatability of dog foods containing different moisture levels, and the inclusion of a mould inhibitor. Animal Feed Science and Technology. 159, 150–155. https://doi.org/10.1016/j.anifeedsci.2010.06.001
Carciofi, A. C., de Oliveira, L. D., Valério, A. G., Borges, L. L., de Carvalho, F. M., Brunetto, M. A., Vasconcellos, R. S. (2009). Comparison of micronized whole soybeans to common protein sources in dry dog and cat diets. Animal Feed Science and Technology. 151(3–4), 251–260. https://doi.org/10.1016/j.anifeedsci.2009.01.002
Cerino, M. C., Castro, D. C., Richard, G. A., Exner, E. L., Pensiero, J. F. (2018). Functional dioecy in Gleditsia amorphoides (Fabaceae). Australian Journal of Botany. 66(2), 85–93. https://doi.org/10.1071/BT16185
Dos Reis, J. S., Zangerônimo, M. G., Ogoshi, R. C. S., França, J., Costa, A. C., Almeida, T. N., Dos Santos, J. P. F., Pires, C. P., Chizzotti, A. F., Leite, C. A. L., Saad, F. M. O. B. (2016). Inclusion of Yucca schidigera extract in diets with different protein levels for dogs. Animal Science Journal. 87: 1019–1027. https://doi.org/10.1111/asj.12535.
Habig, W. H., Pabst, M. J., Jakoby, W. B. (1974). Glutathione S-transferases: The first enzymatic step in mercapturic acid formation. Journal of Biological Chemistry. 249(22), 7130–7139. https://doi.org/10.1016/S0021-9258(19)42083-8
Hoyles, L., Snelling, T., Umlai, U. K., Nicholson, J. K., Carding, S. R., Glen, R. C., McArthur, S. (2018). Microbiome–host systems interactions: protective effects of propionate upon the blood–brain barrier. Microbiome. 6, 55. https://doi.org/10.1186/s40168-018-0439-y
Jiang, Z. Y., Woollard, A. C. S., Wolff, S. P. (1991). Lipid hydroperoxide measurement by oxidation of Fe²⁺ in the presence of xylenol orange: Comparison with the TBA assay and an iodometric method. Lipids. 26(10), 853–856. https://doi.org/10.1007/BF02536169
Lu, G., Ren, T., Zhao, Z., Li, B., Tan, S. (2024). Chemical component differences in the endosperm of Gleditsia species seeds revealed based on comparative metabolomics. Food Chemistry: X, 21, 101060. https://doi.org/10.1016/j.fochx.2023.101060
National Research Council (NRC). (2006). Nutrient requirements of dogs and cats. National Academies Press.
Sedlak, J., Lindsay, R. H. (1968). Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Analytical Biochemistry. 25, 192–205. https://doi.org/10.1016/0003-2697(68)90092-4
Salehian, P., Shareghi, B., Hosseini-Koupaei, M. (2019). Comparative studies on the interaction between biogenic polyamines and bovine intestinal alkaline phosphatases: spectroscopic and theoretical approach. Journal of Biological Physics. 45:89-106. https://doi.org/10.1007/s10867-018-9517-4
Souza, R. B. M. S., Fernandes, E. L., Santos, L. N. A., Lima, L. S., Silva, H. L., Putarov, T. C., Oliveira, S. G., Felix, A. P. (2025). Effects of yeast beta-1,3/1,6-glucans on nutrient digestibility, intestinal functionality, and immune and antioxidant variables in growing dogs submitted to spay or neutering surgery. Plos One. 20(9): e0331843. https://doi.org/10.1371/journal.pone.0331843
Urrego, M. I. G., Matheus, L. F. O., de Melo Santos, K., Ernandes, M. C., Monti, M., de Souza, D. F., Balieiro, J. C. C., Araújo, L. F., Pontieri, C. F. F., Brunetto, M. A. (2017). Effects of different protein sources on fermentation metabolites and nutrient digestibility of brachycephalic dogs. Journal of Nutritional Science. 6, e43. https://doi.org/10.1017/jns.2017.46
Wang, H., Lai, C., Tao, Y., Zhou, M., Tang, R., Yong, Q. (2023). Evaluation of the enzymatic production and prebiotic activity of galactomannan oligosaccharides derived from Gleditsia microphylla. Fermentation. 9(7), 632. https://doi.org/10.3390/fermentation9070632
Zhang, Y., Wang, L., Chen, X., Liu, Y., Li, J. (2023). Quillaja saponaria extract modulates gut microbiota and reduces proteolytic bacteria in dogs: A dose-response study. Veterinary Microbiology. 276, 109634. https://doi.org/10.1016/j.vetmic.2023.109634
Publicado
Como Citar
Edição
Seção
Licença

Este trabalho está licenciado sob uma licença Creative Commons Attribution-ShareAlike 4.0 International License.
Autores que publicam nesta revista concordam com os seguintes termos:
- Autores mantém os direitos autorais e concedem à revista o direito de primeira publicação, com o trabalho simultaneamente licenciado sob a Creative Commons - Atribuição 4.0 Internacional que permite o compartilhamento do trabalho com reconhecimento da autoria e publicação inicial nesta revista.
- Autores têm autorização para assumir contratos adicionais separadamente, para distribuição não-exclusiva da versão do trabalho publicada nesta revista (ex.: publicar em repositório institucional ou como capítulo de livro), com reconhecimento de autoria e publicação inicial nesta revista.
- Autores têm permissão e são estimulados a publicar e distribuir seu trabalho online (ex.: em repositórios institucionais ou na sua página pessoal) a qualquer ponto antes ou durante o processo editorial, já que isso pode gerar alterações produtivas, bem como aumentar o impacto e a citação do trabalho publicado.













